Acids Bases And Salts
Basics of Acids, Bases and Salts
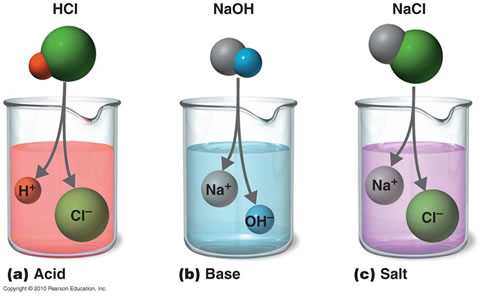
Strength of an acid depends on the concentration of the hydronium ions[H3O+] present in an aqueous solution of an acid. Strength of an Alkali depends on the concentration of hydroxyl ions[OH-] present in an aqueous solution of the alkali.
Read MoreDifference Between Acids and Bases
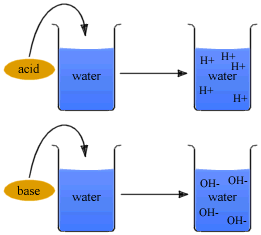
Strength of an acid depends on the concentration of the hydronium ions[H3O+] present in an aqueous solution of an acid. Strength of an Alkali depends on the concentration of hydroxyl ions[OH-] present in an aqueous solution of the alkali.
Read MorePreparation of Acids and Bases

Acids from Salts: Normal + Sulphuric -> Acid +Displaced Salt acid[conc.] salt volatile acid NaCl + H2SO4 NaHSO4 + HCl
Read MorePhysical and Chemical Properties of Acids and Bases

Acids neutralize bases to give salt and water only | Alkalis react with ammonium salts on heating to liberate ammonia.
Read MoreUses of Acids and Bases

Manufacture of soaps, Manufacture of bleaching powder, Food preservation, Baking powder, Cooking
Read More